|
The purpose of the documentation review is to determine whether or not the QMS has been established and documented and meets the established regulatory requirements. Review any applicable standards or guidance documents to refamiliarize yourself with the requirements. Request access to all relevant documents and, if you are auditing a supplier, ask for a map or sketch of their facility that has the departments clearly labeled. Make sure affected members of your organization (or your supplier) understand the scope of the audit you are conducting, when the audit will be conducted, and who is on the team. Establish initial contact with the auditee(s). With a lead auditor chosen, the team determined, the scope defined, and other factors considered, it’s time establish contact with your auditees.That’s why ISO defines competence in terms of education, training, skill, experience, and personal attributes. An auditor who needs to verify process or product measurements may need to have knowledge of quality and statistical tools. Regarding competence, consider this example: An auditor who needs to interview management regarding management processes (e.g., resource processes, results processes, etc.) should have some minimal business experience. The first rule of auditing is that an auditor cannot examine an area for which he/she is responsible. 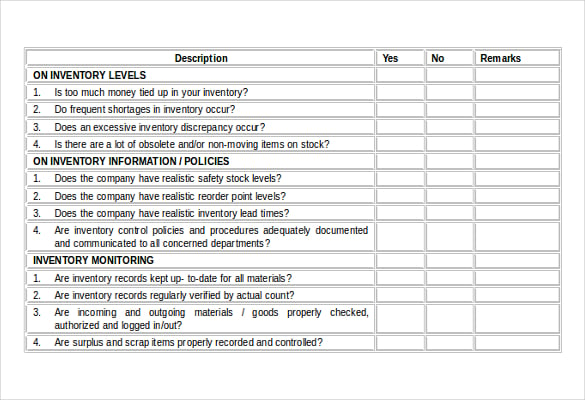
In selecting the audit team members, consider which competencies are needed, how long your audit will last, the scope of the audit, and time constraints. Select the audit team. If your company is small, you may comprise the “team” If your company has more than, say, 150 employees, insources design, makes high risk-products, etc., it is possible that you may need 2+ auditors on your team.Will you have adequate cooperation from auditees? Are any of the people involved working on a major deadline that would take away from their participation? Will any of them be on vacation? Is there adequate time and budget to conduct the audit? Will all the information you need be made available to you? Don’t assume. Determine the feasibility of the audit. You need to ensure that you will be able to conduct the audit as planned.Defining the audit criteria (i.e., ISO 13485:2016) and additional applicable regulatory requirements (e.g., 21 CFR 820 and/or EU Medical Device Regulation 2017/745) are also imperative. You need to define which facilities and/or departments are involved and which processes will be audited and clearly identify those areas that may be excluded. Define audit objectives, scope, and criteria. This is an important step.If you work for a small company, that might be you! This person will be responsible for all phases of the audit. Appoint the lead auditor. The first basic step is to figure out who will lead the audit team.However, the process of initiating the audit is vital to assure the audit process is comprehensive and successful. When planning an audit, it is tempting to skip some of the steps below and go immediately to creating a checklist and schedule. Determine whether or not the QMS has been properly maintained.Determine if the QMS has been effectively implemented. 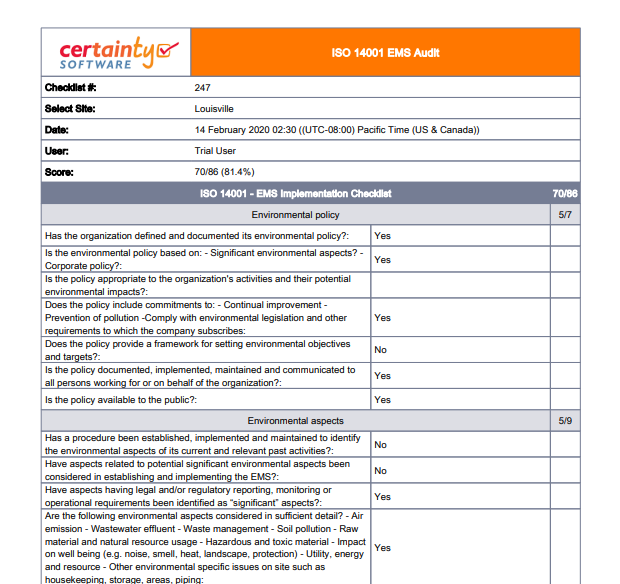
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
